Zoë Jamet et al in Brain
Autoimmune psychiatric disorders represent a fascinating and crucial area of research, shedding light on the intricate interplay between the immune system and neurological function. Among these disorders, anti-NMDA receptor encephalitis stands out due to its severe and diverse symptoms, which include both neurological and psychiatric manifestations. This condition is characterized by the presence of autoantibodies targeting the N-methyl-D-aspartate receptor (NMDAR), leading to significant brain dysfunction.
Recent studies, including investigation from this paper using single-molecule super-resolution imaging techniques on hippocampal neurons, have begun to unravel the complex mechanisms by which NMDAR autoantibodies (NMDAR-Ab) exert their pathogenic effects. In this study, the authors highlight that NMDAR-Ab primarily disrupt extrasynaptic NMDAR, increasing their membrane dynamics and de-clustering their surface interactome within the first few minutes. NMDAR-Ab also induce a rapid reshuffling of all membrane proteins in the extrasynaptic compartment, suggesting a broad and early impact on neuronal cell membrane organization. These early alterations are followed by long-term reductions in synaptic NMDAR pools, characterized by a slowdown in receptor membrane dynamics that occurs independently of cross-linking mechanisms. Notably, exposing only extrasynaptic NMDAR to NMDAR-Ab is sufficient to produce their full-blown effects on synaptic receptors, indicating a cascading impact from extrasynaptic to synaptic sites. Ultimately, these changes impair synaptic transmission and neuronal network properties, contributing to the neurological and psychiatric symptoms observed in patients.
This study demonstrate that NMDAR-Ab first impair extrasynaptic proteins, and then the synaptic ones. These data shed thus new, and unsuspected, lights on the mode of action of NMDAR-Ab and likely to our understanding of (extra)synaptopathies.
Understanding the action of NMDAR-Ab is of paramount importance, not only for comprehending anti-NMDA receptor encephalitis but also for its broader implications in other neurological and psychiatric disorders. The NMDAR is a critical player in various brain functions, and its dysregulation has been implicated in conditions such as Alzheimer’s disease, schizophrenia, and other cognitive and mood disorders. Therefore, studying NMDAR through the lens of these autoantibodies provides valuable insights into the receptor’s role across a spectrum of diseases.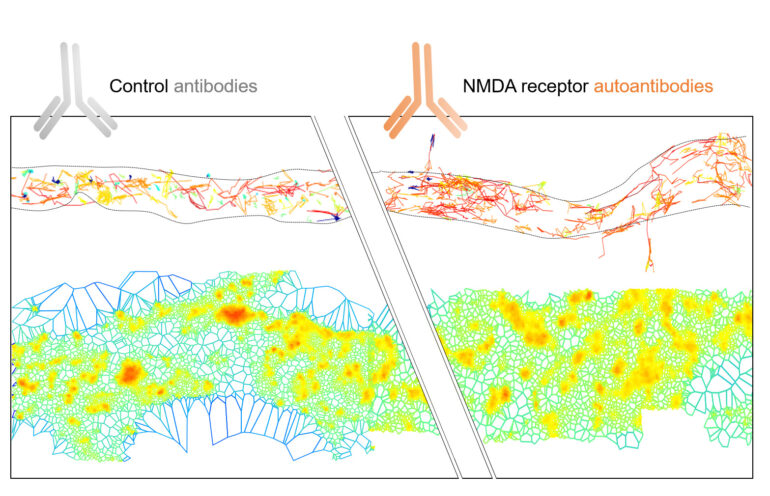
Legend image: Illustration of the diverse mechanism of action of NMDAR autoantibodies (NMDAR-Ab) from patients on membrane proteins. The top section shows the membrane diffusion of NMDAR in the extrasynaptic region of the dendrite with blue indicating less diffusive trajectories and red indicating more diffusive ones. The authors show that extrasynaptic NMDAR surface mobility is acutely affected by NMDAR-Ab with an increased diffusivity (more red trajectories). In the bottom section, super-resolution microscopy (dSTORM) was used to visualize the nano-organization of all surface proteins (surfaceome) in the extrasynaptic region. In the Control-Ab condition (left), the surfaceome is organized into high-density protein clusters (red) along the dendrite. In the presence of NMDAR-Ab (right), the clusters are larger and less dense, indicating that NMDAR-Ab broadly affects all proteins in the extrasynaptic membrane.
Reference
NMDA receptor autoantibodies primarily impair the extrasynaptic compartment
Zoe Jamet 1 , Camille Mergaux 1 , Morgane Meras 1 , Delphine Bouchet 1 , Frédéric Villega 1 2 , Jakob Kreye 3 4, Harald Prüss 3 4 , Laurent Groc 1
1 University of Bordeaux, CNRS, Interdisciplinary Institute for Neuroscience, IINS, UMR 5297, F-33000, Bordeaux, France.
2 Department of Pediatric Neurology, CIC-0005, University Children’s Hospital of Bordeaux, F-33000, Bordeaux, France.
3 German Center for Neurodegenerative Diseases (DZNE) Berlin, 10117 Berlin, Germany.
4 Department of Neurology and Experimental Neurology, Charité-Universitätsmedizin Berlin, Corporate Member of Freie Universität Berlin, Humboldt-Universität Berlin, 10117 Berlin, Germany.
Brain. 2024 May 17:awae163.
https://doi.org/10.1093/brain/awae163
Last update 16/04/25
